
Order the NovaSeq 6000Dx Instrument
The NovaSeq 6000Dx instrument offers high-throughput sequencing in either IVD or RUO Mode, with accelerated data analysis.
A library preparation and enrichment solution for IVD targeted sequencing applications.
This product is only available in select locations. Please select the location where you would like this product to be shipped to see availability.
Illumina DNA Prep with Enrichment Dx enables clinical labs to add targeted sequencing enrichment panels to their diagnostic applications and is an approved IVD medical device in the United States, the European Union, the United Kingdom, and other international markets.
This FDA-registered and EU IVDR 2017/746–compliant library prep and enrichment kit enables clinical labs to add targeted sequencing enrichment panels to their diagnostic applications.
The kit is compatible with FDA-registered and CE-marked IVD NGS platforms that use proven sequencing by synthesis (SBS) chemistry to deliver highly accurate diagnostic testing.
Local Run Manager in Dx mode offers onboard primary analysis with a user-friendly interface on the MiSeqDx or NextSeq 550Dx instruments. The DRAGEN for Illumina DNA Prep with Enrichment Dx App performs alignment and variant calling in DNA for somatic and germline mutations. This app automatically launches secondary analysis when paired with an On-Premise DRAGEN Server and the NextSeq 550Dx or NovaSeq 6000Dx instruments via Illumina Run Manager.
Illumina DNA Prep with Enrichment Dx Kit is a set of reagents and consumables used to prepare sample libraries from genomic DNA derived from human cells and tissue to develop in vitro diagnostic assays. User supplied probe panels are required for the preparation of libraries targeting specific genomic regions of interest. The generated sample libraries are intended for use on Illumina sequencing systems. The Illumina DNA Prep with Enrichment Dx includes software for sequencing run setup, monitoring, and analysis.
Illumina DNA Prep with Enrichment Dx is a set of reagents and consumables used to prepare sample libraries from genomic DNA derived from human cells and tissue. User-supplied probe panels are required for the preparation of libraries targeting specific genomic regions of interest. The generated sample libraries are intended for use on Illumina sequencing systems.
| Assay time | ~7 hours |
|---|---|
| Hands-on time | ~1 hr |
| Input quantity | 50-1000 ng genomic DNA derived from human cells and tissue including whole blood or FFPE tissue |
| Instruments | NovaSeq 6000Dx Instrument, MiSeqDx Instrument, NextSeq 550Dx Instrument |
| Method | Custom sequencing, Targeted DNA sequencing, Exome sequencing, Target enrichment |
| Nucleic acid type | DNA |
| Species category | Human |
| Technology | Sequencing |
To prepare targeted NGS libraries, Illumina DNA Prep with Enrichment Dx requires a user-supplied compatible enrichment panel and Local Run Manager. Speak with an Illumina sales representative to arrange installation on your Dx instrument.
Instrument-specific reagent kits are also required: MiSeqDx Reagent Kit v3 (Catalog no. 20037124), NextSeq 550Dx High Output Reagent Kit v2.5 (300 cycles) (Catalog no. 20028871), or NovaSeq 6000Dx Reagent Kit v1.5 (300 Cycles) (Catalog no. 20046931 or 20046933).
Illumina DNA Prep with Enrichment Dx is an approved and registered IVD library prep and enrichment solution for in vitro diagnostic applications on the MiSeqDx, NextSeq 550Dx, and NovaSeq 6000Dx Instruments.
Illumina DNA Prep with Enrichment Dx
DRAGEN for Illumina DNA Prep with Enrichment Dx App**
*Local Run Manager in Dx mode offers onboard primary analysis on the MiSeqDx or NextSeq 550Dx instruments.
**Automatically launches when paired with an On-Premise DRAGEN Server and the NextSeq 550Dx or NovaSeq 6000Dx instruments via Illumina Run Manager.
High-quality variant calling with the NovaSeq 6000Dx instrument
Access a validated workflow for highly accurate germline and somatic variant calling.
NextSeq 550Dx applications and methods
Explore the versatility of application workflows for developing IVD tests and a range of research methods on a single instrument.
MiSeqDx applications and methods
MiSeqDx applications include IVD approved and registered assays for cystic fibrosis testing and NGS-based assay development.
In vitro diagnostic (IVD) instruments
Explore IVD instruments designed for the clinical lab environment.
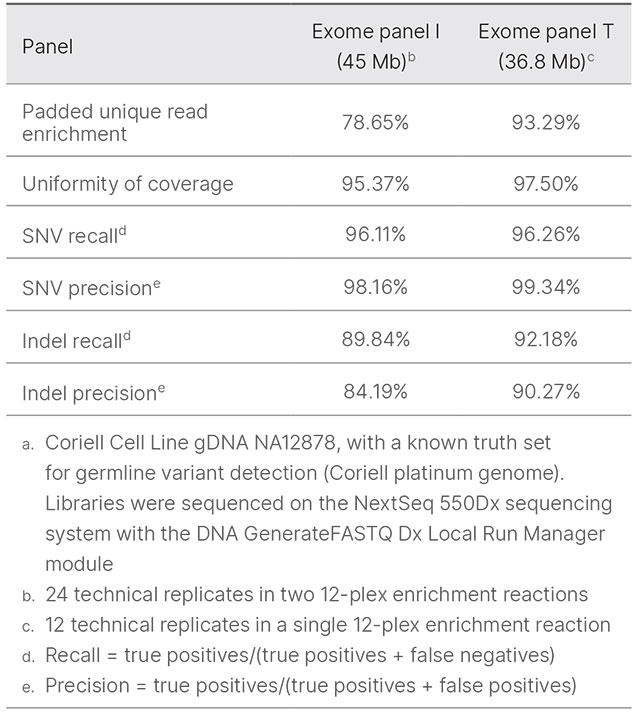

a. Coriell Cell Line gDNA NA12878, with a known truth set for germline variant detection (Coriell platinum genome). Libraries were sequenced on the NextSeq 550Dx Instrument with FASTQ files generated from base calls using the GenerateFASTQ Dx Module in Local Run Manager; custom scripts in the DRAGEN platform v3.8.4 were used for analysis.
Illumina DNA Prep with Enrichment Dx with UD Indexes Set A (16 samples)
20051354
CE-marked and FDA-regulated IVD reagents , including index set A and size selection beads, for library preparation of genomic DNA isolated from 16 FFPE tissue samples. Purchase MiSeqDx, NextSeq 550Dx, or NovaSeq 6000Dx sequencing reagents separately.
List Price:
Discounts:
Illumina DNA Prep with Enrichment Dx with UD Indexes Set B (16 samples)
20051355
CE-marked and FDA-regulated IVD reagents , including index set B and size selection beads, for library preparation of genomic DNA isolated from 16 FFPE tissue samples. Purchase MiSeqDx, NextSeq 550Dx, or NovaSeq 6000Dx sequencing reagents separately.
List Price:
Discounts:
Illumina DNA Prep with Enrichment Dx with UD Indexes Set A (96 samples)
20051352
CE-marked and FDA-regulated IVD reagents , including index set A and size selection beads, for library preparation of genomic DNA isolated from 96 peripheral whole blood samples. Purchase MiSeqDx, NextSeq 550Dx, or NovaSeq 6000Dx sequencing reagents separately.
List Price:
Discounts:
Illumina DNA Prep with Enrichment Dx with UD Indexes Set B (96 samples)
20051353
CE-marked and FDA-regulated IVD reagents , including index set B and size selection beads, for library preparation of genomic DNA isolated from 96 peripheral whole blood samples. Purchase MiSeqDx, NextSeq 550Dx, or NovaSeq 6000Dx sequencing reagents separately.
List Price:
Discounts:
DNA Prep Enrichment Dx-Customer Site
20076400
1.5-day, hands-on instructions at customer site to familiarize users with Illumina DNA Prep with Enrichment Dx library prep workflow. Course provides hands-on sample and library prep, enrichment, quality control, best practices, troubleshooting, and related Illumina-supported sequencing and data analysis tools for a maximum of four trainees.
TruSight Whole Genome Customer Training – Customer Site
20122846
A 1.5-day onsite training for the complete TruSight Whole Genome Dx Library Preparation workflow, with detailed hands-on instruction. Participants will learn all steps of the workflow including sample and library preparation, quality control, best practices, and troubleshooting. Training on Illumina-supported sequencing and data analysis tools specific to the TruSight Whole Genome workflow will be included.
Showing of
Product
Qty
Unit price
Product
Catalog ID
Quantity
Unit price
Illumina DNA Prep with Enrichment Dx uses innovative on-bead tagmentation, which uses bead-bound transposomes to mediate a uniform reaction that integrates DNA extraction, fragmentation, library prep, and library normalization. This provides a rapid library prep and enrichment solution when combined with a simplified hybridization step. The kit includes clean-up beads for library purification and sequencing indexes.
You will need to order the Dx reagents designated for your Illumina Dx sequencing Instrument:
Not all reagent kits will be available in all countries. Please visit each product page to learn more.
Illumina DNA Prep with Enrichment Dx supports a wide range of genomic DNA (gDNA) derived from cells and tissue. This includes gDNA extracted from whole blood or formalin-fixed, paraffin-embedded (FFPE) tissue.
Have additional questions about Illumina DNA Prep with Enrichment Dx? Contact us today.
Your email address is never shared with third parties.