TruSight Whole Genome Assay
An EU IVDR 2017/746-compliant, scalable whole-genome sequencing solution for clinical diagnostic testing.
Overview
Key features
Specifications
| Method | Whole-Genome Sequencing |
|---|---|
| Nucleic acid type | DNA |
| Species category | Human |
| Systems | NovaSeq 6000Dx Instrument |
| Technology | Sequencing |
Required Products
Applications
Example workflow
Project recommendations
| Instrument | Recommended number of samples | Read length |
|---|
Related applications and methods
Documentation
Product literature
Literature is not currently available for this product.
Support documentation
A dedicated support section is not currently available for this product
Compare
Selection tools:
Figures
TruSight Whole Genome Assay
TruSight Whole Genome Assay
TruSight Whole Genome Assay
Resources
TruSight Whole Genome Assay
TruSight Whole Genome Assay
TruSight Whole Genome Assay
TruSight Whole Genome Manual (24 sample)
20093209
TruSight Whole Genome Manual (24 sample)
NovaSeq 6000Dx S2 Reagent Kit v1.5 (300 cycles) IVD
20046931
Includes one S2 flow cell (with four lanes), one cluster cartridge, and one sequencing by synthesis (SBS) cartridge to support one 300-cycle run on the NovaSeq 6000Dx. Requires separate purchase of one buffer cartridge and one library tube for each run. All products registered for In Vitro Diagnostic sequencing.
List Price:
Discounts:
NovaSeq 6000Dx S2 Buffer, IVD
20062292
Includes one S2 buffer reagent cartridge to dilute denatured libraries prior to sequencing, required to support each S2 flow cell 300-cycle run.
List Price:
Discounts:
NovaSeq 6000Dx S4 Buffer Cartridge
20062293
Includes one S4 buffer reagent cartridge to dilute denatured libraries prior to sequencing, required to support each S4 flow cell 300-cycle run.
List Price:
Discounts:
NovaSeq 6000Dx Library Tube, IVD
20062290
Includes one NovaSeq 6000Dx Library Tube, IVD, required for each S2 or S4 flow cell 300-cycle run.
List Price:
Discounts:
NovaSeq 6000Dx Library Tubes Accessory Pack (24 tubes) IVD
20062291
Includes 24 NovaSeq 6000Dx Library Tubes.
List Price:
Discounts:
Showing of
Selection summary
Product
Qty
Unit Price
Items added to the cart
Summary
Product
Catalog ID
Quantity
Unit price
-
Catalog ID:Qty:
FAQs
Speak with a specialist
Reach out for information about our products and services, or get answers to questions about our technology.
Your email address is never shared with third parties.
Product Highlights
The TruSight Whole Genome Assay enables clinical laboratories to add WGS to their menu of diagnostic applications and offers:
- A comprehensive, DNA-to-VCF data file workflow
- PCR-free library preparation chemistry for minimal bias and superior coverage uniformity compared to targeted approaches
- Optimal performance with included library QC metrics and filtering, without the need for external batch controls
The TruSight Whole Genome offers an EU IVDR–compliant and comprehensive, DNA-to-variant call format (VCF) workflow solution for clinical WGS. This validated solution facilitates adoption of WGS for clinical applications by removing the burdens of developing controls, building bioinformatic pipelines, and performing costly and time-consuming analytical validation studies.
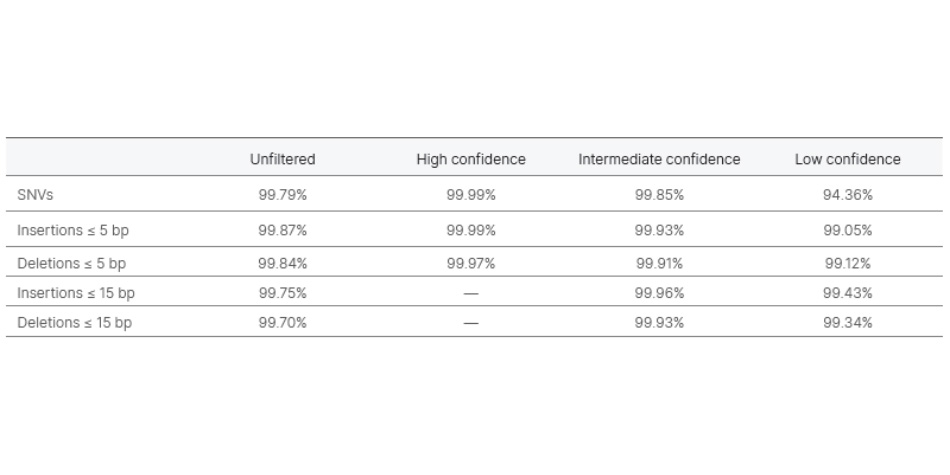
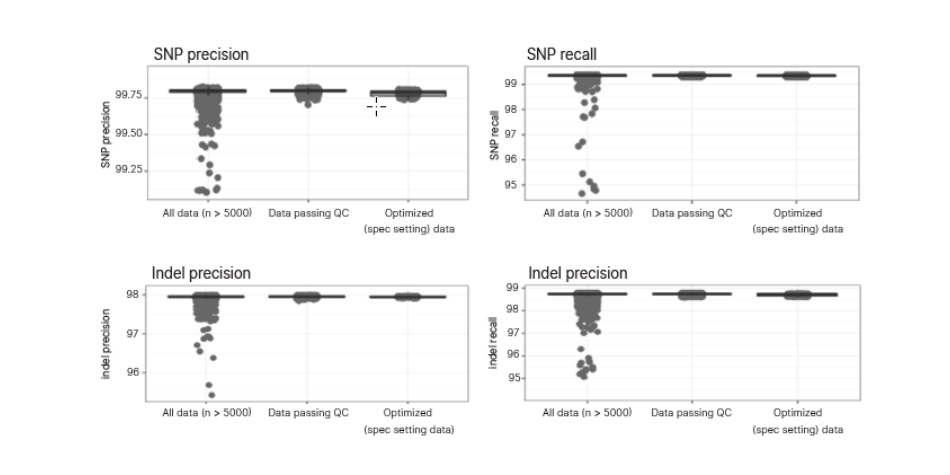
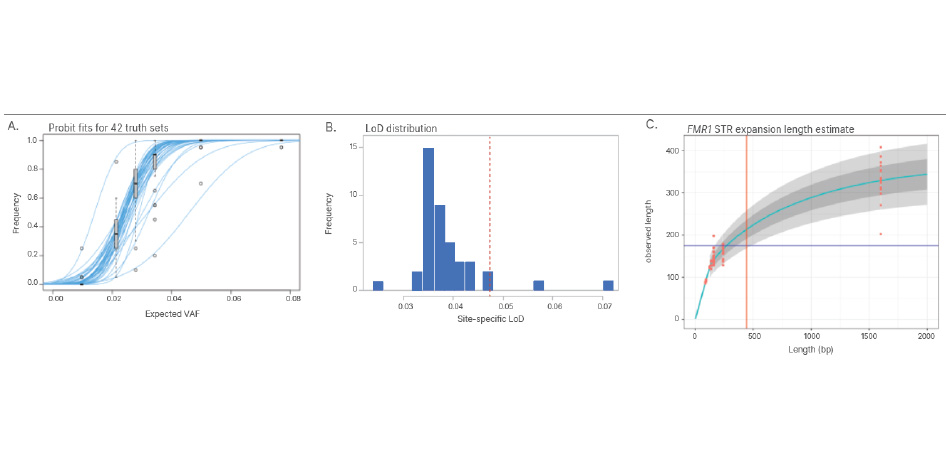
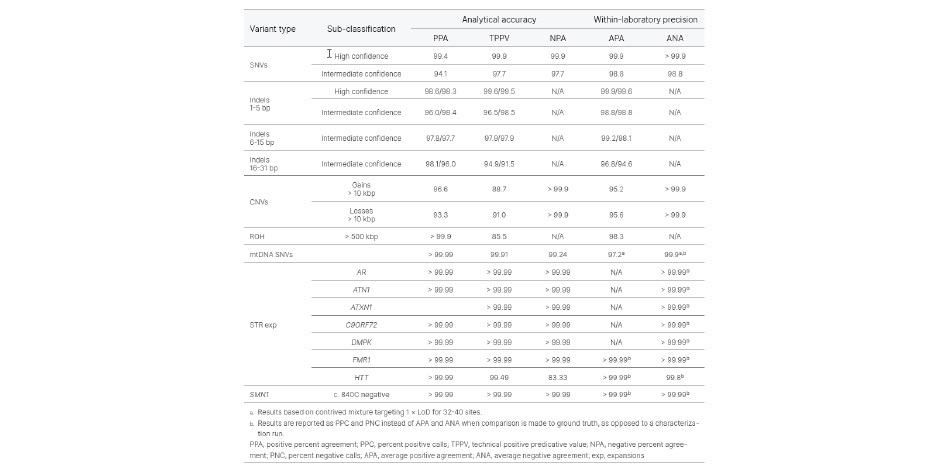
Analytical validation of the TruSight Whole Genome Assay was performed in accordance with Clinical and Laboratory Standards Institute (CLSI) guidelines with over 450 clinical samples and over 300 sequencing runs. QC metrics and confidence regions included in the development of the assay identify poor performing samples and filter data. This reduces false-positive and false-negative calls and significantly improves performance.
Library Preparation:
TruSight Whole Genome Library Prep
Sequencing:
Data Analysis:
TruSight Whole Genome Analysis App via DRAGEN server with Illumina Run Manager*
* Must be installed by a Field Service Representative on the NovaSeq 6000Dx instrument
Specifications
The TruSight Whole Genome Assay requires installation of the TruSight Whole Genome Analysis Application on your NovaSeq 6000Dx instrument by a Field Service Representative. Contact your sales or field representative to schedule this installation prior to running the assay.
Intended Use
The TruSight Whole Genome Assay is a qualitative in vitro diagnostic device intended for whole-genome sequencing and detection of copy number variants, single nucleotide variants, runs of homozygosity, insertion/deletions, short tandem repeat expansions, and mitochondrial variations in human genomic DNA extracted from blood.
The TruSight Whole Genome Assay includes the TruSight Whole Genome Library Prep Kit and the TruSight Whole Genome Application Software. The device is intended to be used with the NovaSeq 6000Dx Instrument and downstream diagnostic germline analysis applications compatible for use with this device.